Abbott Laboratories is accused in a trial that started Tuesday of marketing unapproved stents for unsuspecting patients and cheating Medicare, as a whistleblower seeks more than $1 billion, a risk the company hasn’t fully disclosed to shareholders.
The company faces allegations it increased sales by marketing devices for uses that weren’t approved by the U.S. Food and Drug Administration. Kevin Colquitt, a former salesman for Abbott and Guidant LLC turned whistleblower, claims it pushed bile duct stents, intended for short-term purposes, for more complex vascular use.
The drugmaker didn’t conduct a clinical trial on the safety, instead using so-called off-label marketing to test devices on patients without their informed consent, Colquitt claims. Multiple patients suffered complications including fractures of the devices after implantation, stroke, infections and migration of the stents, he alleges.
Abbott, which acquired Guidant’s stent business in 2006, denies the claims and calls the lawsuit an attempt to “rewrite history.” The use of biliary, or bile duct, stents to treat peripheral vascular or arterial disease was “a generally accepted medical practice and indeed, standard of care,” Abbott lawyers said in court papers.
Doctors used the stents to provide “best patient care” and Medicare knowingly approved payments, the company said.
Jury Trial
The three-week federal court jury trial in Dallas covers some of the same products at issue in a $5.5 million settlement Abbott reached with the U.S. in 2013 over kickbacks paid to doctors to use the stents, which are made to be inserted in bile ducts to keep them open. The Dallas suit focuses on off-label marketing, the practice of actively marketing a drug or device for an unapproved use.
The case may turn simply on what jurors think of claims that patients weren’t told the devices hadn’t been approved, said Patrick Burns, of Taxpayers Against Fraud, a Washington advocacy group.
“There is no need to prove that any individual was harmed,” he said in an interview. “But who would allow bile duct stents to be put in themselves or their mother or father if they weren’t FDA approved?” he asked. “If Abbott loses this case, they’re going to lose it big.”
Court Rulings
U.S. District Judge Barbara Lynn in January rejected Abbott’s request to throw out the case. She also denied Colquitt’s motion to find Abbott liable for making false claims before trial.
Guidant “pursued a reimbursement-driven marketing strategy for off-label use of biliary stents that were not only unapproved for vascular indications, but carried an express warning from the FDA that the devices were not established to be safe or effective,” Colquitt’s lawyers said in an October filing.
The biliary stents weren’t eligible for Medicare reimbursement because they weren’t approved for vascular use, according to the lawsuit. About 99 percent of the bile duct stents the company sold from 2004 to 2006, the time period covered by the lawsuit, were used off-label, according to testimony by an Abbott expert, court papers show.
Guidant targeted doctors who normally didn’t use bile duct stents in order to increase sales for a product that had limited market prospects, according to the lawsuit.
Thousands of Patients
“Several thousand patients are diagnosed annually with terminal biliary cancer. Life expectancy is limited,” Colquitt’s lawyers said. “Hundreds of thousands of patients annually require vascular stents to treat peripheral vascular disease.”
In one 2002 e-mail, cited in court papers, a Guidant executive noted a company lawyer “expressed concerns over our training and sale of this product so far off label.” According to this e-mail, referring to internal discussions, “There was some serious concern about a paper trail of data on a device for biliary applications handed to a sales force that does not do biliary interventions.”
Abbott coached doctors and other health-care providers on how to use billing codes to get past the Medicare reimbursement requirement, Colquitt alleges. Each claim for reimbursement –- thousands of them — was a false statement, according to the lawsuit.
Cost Estimate
Colquitt’s damages expert estimated that off-label promotion caused the federal government to pay $214.7 million to $330.4 million for peripheral vascular stent procedures using unapproved stents.
Colquitt filed the lawsuit under the federal False Claims Act, allowing any damages awarded by a jury to be tripled. The money would go to the U.S., with Colquitt eligible for a share. The lawsuit also seeks fines of from $5,500 to $11,000 for each of the alleged false claims.
Abbott denies claims that Medicare was fooled into paying for the stents or that such payments were barred.
“The government entities implementing the Medicare program knew all about the widespread, well-accepted use of biliary stents in vascular stenting procedures and chose to reimburse such procedures,” Abbott’s lawyers wrote in a September filing. “This case involves medical treatment using devices that were indisputably standard-of-care in procedures the government deliberately (and correctly) exercised its discretion to reimburse.”
Blood Flow
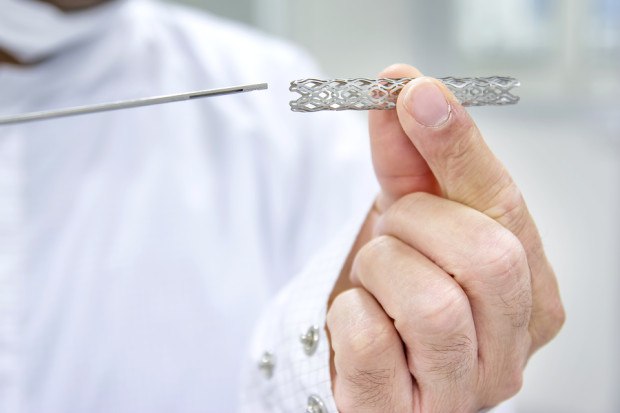
The stents, small tubular metal objects, would be placed in arteries to increase blood flow, generally for patients with peripheral arterial or peripheral vascular disease. “If left untreated, PAD can cause gangrene, loss of a limb, renal failure, and even death,” Abbott said in court papers. Using a stent is a safer alternative treatment to invasive surgery, the company said.
Even if Colquitt could establish a false claim, Abbott lawyers argued, “he cannot establish Guidant knowingly caused its submission or knowingly made any false statements material to a false claim.” The U.S. law requires that a defendant was knowingly or recklessly responsible for a false claim to be held liable. Guidant believed off-label use of the stents was reimbursable — “just like everyone else at the time,” the company said.
Biliary stents were approved by the U.S. Food and Drug Administration to temporarily drain the biliary tract when cancer or another obstruction has blocked the flow of bile that the body produces to help digestion, or to prevent the bile duct from crimping.
Blood Vessels
Doctors over the years used them in other areas such as the blood vessels that run down the legs since they were larger than the stents used to prop open heart arteries.
Millennium Research Group, a market research firm, estimated that stent sales for the approved biliary use were $37.6 million in 2006. More than 1 million more devices were implanted in other non-approved areas, called off-label use, between 2003 and 2006, according to a study published in the American Journal of Therapeutics in February 2008. The same year, Johnson & Johnson, which also made the stents, estimated the market would reach $1.8 billion by 2012.
While doctors are allowed to use devices without formal FDA approval, the agency warned them and device makers about the risks after receiving reports of hundreds of adverse events. An FDA analysis found more than 90 percent of biliary stent injuries and malfunctions between 2001 and 2006 occurred when the devices were used in off-label procedures.
Almost all the patients were elderly and attorneys with the AARP Foundation litigation unit joined the lawsuit in 2010 as co-counsel against Abbott. Kelly Bagby, an AARP attorney; Chris Hamilton, Colquitt’s lawyer; and Darcy Ross, an Abbott spokeswoman, declined to comment on the case before the trial.
The case is U.S. ex rel. Colquitt v. Abbott Laboratories f/k/a Guidant Corp., 06-cv-01769, U.S. District Court, Northern District of Texas (Dallas).




















 How We’re Doing It: Customers, Agents Play Vital Roles in Commercial Auto Telematics Success
How We’re Doing It: Customers, Agents Play Vital Roles in Commercial Auto Telematics Success  From ‘FBI Claims Handling’ to AI-Assisted Workflows
From ‘FBI Claims Handling’ to AI-Assisted Workflows  The Financial Case for Negotiation: How Indemnity Discipline Can Transform Carrier Economics
The Financial Case for Negotiation: How Indemnity Discipline Can Transform Carrier Economics  Storm Counts, Landfalls and Losses: The Hidden Risk Behind a ‘Quiet’ Hurricane Season
Storm Counts, Landfalls and Losses: The Hidden Risk Behind a ‘Quiet’ Hurricane Season 









